
Bioanalytical testing laboratory specializing in large molecule bioanalysis.

INVESTMENT | 2018 (partial exit in 2021)
OWNERSHIP | Minority

Performance 2025
In 2025, BioAgilytix continued to strengthen its platform and invest in capabilities that support customers across the full drug development lifecycle. The company launched state-of-the-art laboratory facilities across multiple geographies, broadening its service offerings and introducing new capabilities.
Additionally, BioAgilytix strengthened the team with strategic hires and rolled out a new Electronic Lab Notebook system to improve automation, data quality, and efficiency.
Commercially, BioAgilytix achieved a record level of new customer bookings, despite an uncertain biopharma funding environment. Performance with large biopharma customers was particularly strong, with the company making meaningful inroads with blue chip pharmaceutical clients.
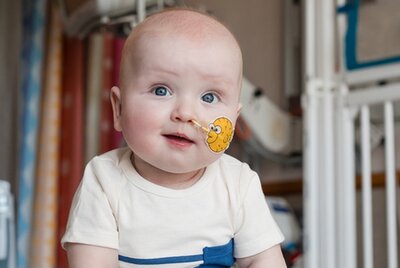
Enabling a Medical First
In 2025, BioAgilytix contributed to a major medical first by supporting the development of the world’s first personalized in vivo CRISPR gene editing therapy, administered to Baby KJ, an infant with a rare and life threatening genetic disease. The therapy was designed, tested, and delivered in record time for a single patient, marking a breakthrough in personalized genetic medicine and highlighting BioAgilytix’s role in enabling cutting-edge innovation in advanced therapies.

Durham, USA | HQ
+1,100 | Employees
6 | Locations
